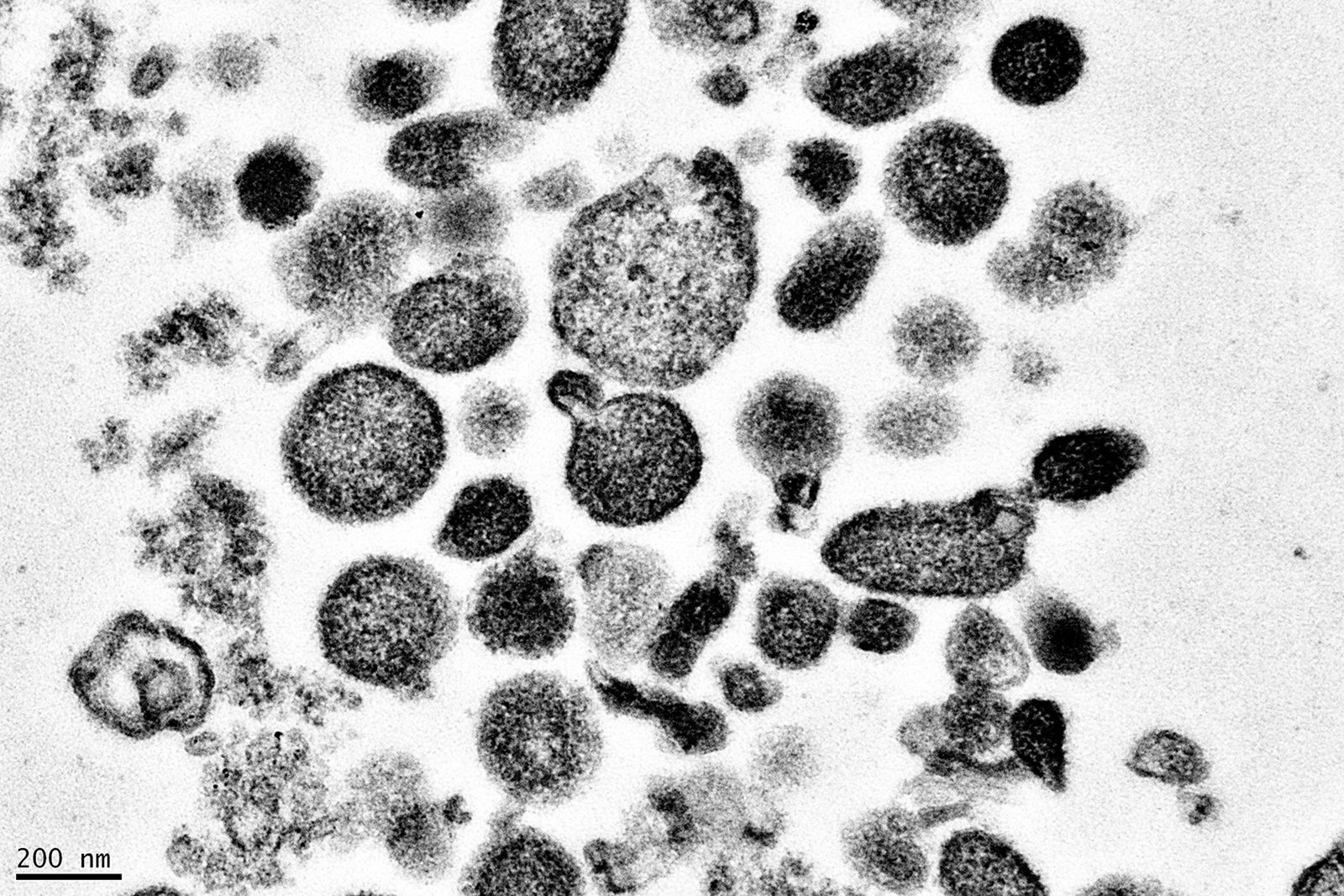
Extracellular Vesicles and Biotherapy Development
Long considered uninteresting cellular debris, it is now appreciated that extracellular vesicles (EVs) can play a major role in intercellular signaling. EVs can arise from endogenous production pathways (exosomes) or bud from the cell surface (ectosomes, or microvesicles). These particles can be harnessed by viruses to deliver immune signals to cells in trans. In addition, EVs are generated in blood products, and these EVs can modulate immune responses in cells exposed to the EVs.
Our group measured the evolution of EV subtypes in stored blood and determined the influence of blood manufacturing method on EV generation. In addition, we found that EVs in blood interact primarily with monocytes, inducing a pro-inflammatory response in these cells. The immunologically active portion of EVs was contained in the smaller, exosome fraction. More recent studies of critically ill subjects has led to the development of a model whereby EVs generated by endothelial cells, granulocytes, and platelets activate monocytes, which can lead to increased tissue damage.
Our group has also made significant contributions into methods to quantitate and characterize EVs by flow cytometry. We defined methods to avoid contamination by pre-filtering fluorochrome-conjugated antibodies to remove aggregates and to decrease background signal through washing and gating strategies. These techniques are now being applied to subjects in clinical trials of blood transfusion to detect immune modulation induced by transfusion. Current efforts include modifying EV-producing cells to change the ability of EVs to modulate target cells.